Mutat Res 204:307–316Īruldhas MM, Subramanian S, Sekar P, Vengatesh G, Chandrahasan G, Govindarajulu P, Akbarsha M (2005) Chronic chromium exposure-induced changes in testicular histoarchitecture are associated with oxidative stress: study in a non-human primate (Macaca radiata Geoffroy). The Collaborative Study Group for the Micronucleus Test. Reprod Fertil Dev 10:179–184Īnonymous (1988) Strain difference in the micronucleus test. Int J Pharm Technol 8:18676–18684Īl-Hamood M, Elbetieha A, Bataineh H (1998) Sexual maturation and fertility of male and female mice exposed prenatally and postnatally to trivalent and hexavalent chromium compounds. This article reviews the toxicological effects of Cr(VI) and Cr(III) and their mechanisms of toxicity and carcinogenicity.Īhmadi S, Rastegar S, Makhdoumi P, Hossini H (2016) Removal of hexavalent chromium from aqueous solution using polyaniline: modeling and optimization. Chromium pollution can have severe consequences for water and the soil environment. Chronic exposure and bioaccumulation of chromium, as a heavy metal, can cause toxicity and numerous pathophysiological defects, including allergic reactions, anemia, burns, and sores especially in the stomach and small intestine, damage to sperm along with the male reproductive system, and affect various biological systems. Considerably, Cr(VI) is frequently more toxic than Cr(III) because of its particular solubility and high mobility. At among, trivalent and hexavalent chromium are the most stable forms. Chromium has been found in the environment in different oxidation states such as Cr 0, Cr(III), and Cr(VI) and is released from a variety of anthropogenic and natural activities. 2016).The growing use of heavy metals in most industrial activities has led to it being considered as the most important environmental pollutant that may cause harm and toxicity to animals and humans. OH according to Equations ( 1) and ( 2), which can be treated as the mechanism of Fenton-like reaction ( Rahim Pouran et al.H 2O 2 reacts with transition metal ions to generate Hydrogen peroxide (H 2O 2) is widely used in the treatment of wastewater as an oxidant like the Fenton-like method ( Li et al. 2017a), but the problems associated with the high temperature salt roasting technologies, including the low overall resource utilization efficiency, the high energy consumption, and the environmental pollution (toxic gas, and waste water), still remain unsolved. 2017b), and hydrometallurgical methods such as pressure oxidative leaching coupled with oxygen, ozone, permanganate and ferrate have been developed ( Zhang et al. 
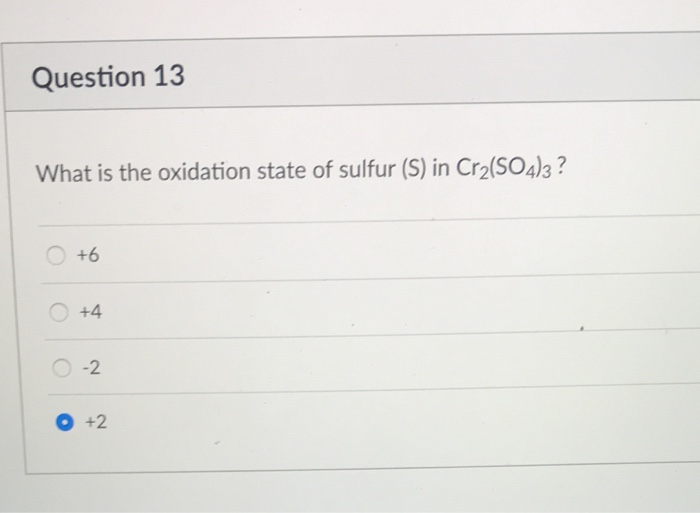
Conventionally, sodium roasting-water leaching technology, calcium roasting-acid leaching ( Xue et al. Many hydrometallurgical processes have been proposed to recover chromium ( Shiyuan et al. Oxidation efficiency was nearly 100% under the optimal conditions: volume ratio of H 2O 2 to mass of Cr 2(SO 4) 3 of 2.4 mL/g, mass ratio of NaOH to Cr 2(SO 4) 3 0.6 g/g, reaction time of 90 min, reaction temperature of 90 ☌ and stirring rate of 500 rpm. The oxidation efficiency was significantly affected by the dosage of H 2O 2 and NaOH, reaction time and reaction temperature took second place last was the stirring rate. The effect of parameters including dosage of H 2O 2, dosage of NaOH, reaction time, reaction temperature and stirring rate on the oxidation efficiency of chromium were investigated. This paper focuses on the oxidation process of chromium (III) with hydrogen peroxide (H 2O 2) in an alkaline medium. Many technologies have been proposed to oxidize chromium, such as roasting-water leaching technology and hydrometallurgical methods such as pressure oxidative leaching coupled with oxygen, ozone, permanganate and ferrate, but the problems associated with the high temperature, low overall resource utilization efficiency, high energy consumption, and the environmental pollution, still remain unsolved.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
